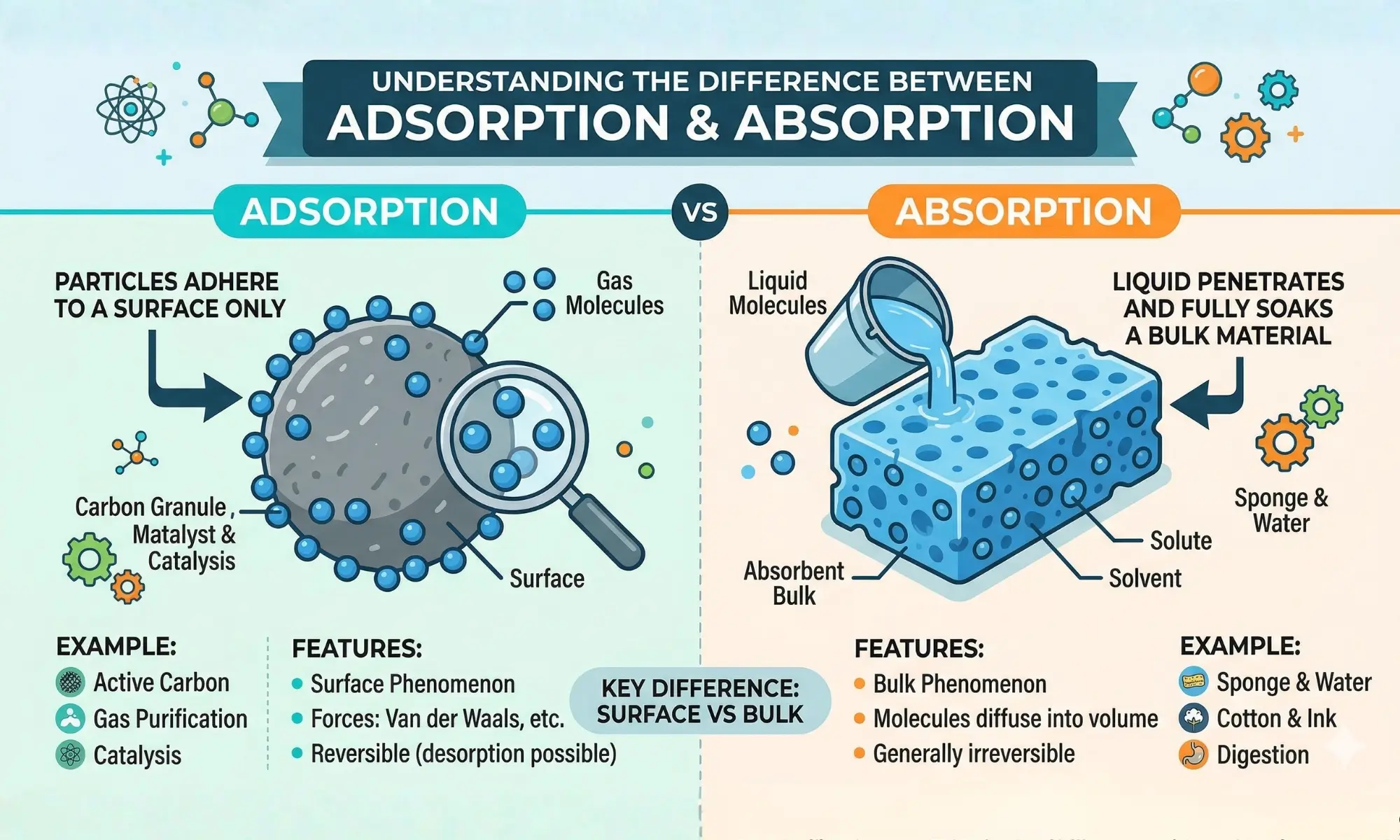
Imagine you spill water on a sponge and also notice dust sticking to a glass surface. Both situations look similar, but they are not the same. This is where the difference between adsorption and absorption becomes important.
Adsorption is when particles stick to a surface, while absorption happens when a substance is fully soaked inside another.
Understanding the difference between adsorption and absorption helps students, scientists, and even everyday learners explain common real-life processes. For example, air purifiers use adsorption, while sponges show absorption.
The difference between adsorption and absorption is often confusing, but once you see real examples, it becomes easy to understand.
In this guide, we will clearly explain the difference between adsorption and absorption in simple words so anyone can learn it quickly.
Key Difference Between Adsorption and Absorption
- Adsorption: Substance sticks only on the surface.
- Absorption: Substance spreads throughout the whole material.
Why Is Their Difference Important?
Knowing the difference between adsorption and absorption is very useful in science and daily life. Students need it to understand chemistry topics like surface reactions. Experts use it in industries like water purification, medicine, and gas masks.
For example, adsorption helps remove pollutants from air, while absorption helps medicines enter the body. This knowledge improves technology, health, and environmental safety. Without understanding their difference, people may use the wrong method in real-life applications.
Pronunciation
- Adsorption
- US: /ædˈsɔːrpʃən/
- UK: /ədˈsɔːpʃən/
- Absorption
- US: /əbˈzɔːrpʃən/
- UK: /əbˈzɔːpʃən/
Now that you know the basics, let’s explore the detailed differences.
Difference Between Adsorption and Absorption
1. Definition
- Adsorption: Particles stick to the surface.
- Example: Dust sticking on a table.
- Example: Gas sticking on charcoal.
- Absorption: Particles enter fully inside.
- Example: Water soaking into a sponge.
- Example: Ink absorbed by paper.
2. Nature
- Adsorption: Surface process.
- Example: Smoke particles on walls.
- Example: Pollutants on activated carbon.
- Absorption: Bulk process.
- Example: Water in soil.
- Example: Sugar dissolving in water.
3. Concentration
- Adsorption: Higher on surface.
- Example: Gas layer on metal.
- Example: Oil on water surface.
- Absorption: Same throughout.
- Example: Salt in water.
- Example: Juice in sponge.
4. Speed
- Adsorption: Fast at first, then slows.
- Example: Air filter trapping dust quickly.
- Example: Charcoal absorbing gas initially.
- Absorption: Steady process.
- Example: Water entering cloth slowly.
- Example: Chemical dissolving in liquid.
5. Heat Effect
- Adsorption: Releases heat.
- Example: Gas on solid surface.
- Example: Cooling systems.
- Absorption: May absorb heat.
- Example: Dissolving salts.
- Example: Chemical mixing.
6. Reversibility
- Adsorption: Easily reversible.
- Example: Gas removed by heating.
- Example: Cleaning filters.
- Absorption: Hard to reverse.
- Example: Water in sponge.
- Example: Dye in fabric.
7. Dependence
- Adsorption: Depends on surface area.
- Example: Powder works better than solid block.
- Example: Activated charcoal.
- Absorption: Depends on volume.
- Example: Bigger sponge absorbs more.
- Example: Large liquid containers.
8. Example Materials
- Adsorption: Charcoal, silica gel.
- Example: Air purifiers.
- Example: Gas masks.
- Absorption: Sponges, liquids.
- Example: Towels.
- Example: Soil absorbing water.
9. Energy
- Adsorption: Low energy process.
- Example: Weak attraction forces.
- Example: Surface bonding.
- Absorption: Higher energy changes.
- Example: Chemical mixing.
- Example: Liquid reactions.
10. Applications
- Adsorption: Purification, catalysis.
- Example: Water filters.
- Example: Industrial cleaning.
- Absorption: Digestion, chemical solutions.
- Example: Nutrients in body.
- Example: Medicine intake.
Nature and Behavior
- Adsorption: Works only on the outer surface and depends on contact area.
- Absorption: Works inside the material and spreads evenly.
Why People Get Confused
People confuse them because both involve one substance interacting with another. Also, the words sound similar, which adds to the confusion.
Comparison Table
| Feature | Adsorption | Absorption | Similarity |
| Process | Surface | Bulk | Both involve substances |
| Speed | Fast initially | Steady | Both time-dependent |
| Heat | Releases heat | May absorb heat | Energy involved |
| Reversibility | Easy | Difficult | Can be controlled |
Which Is Better in What Situation?
Adsorption is better when you need to remove unwanted particles from surfaces, like air purification or water cleaning. It works quickly and is easy to reverse.
Absorption is better when you need a substance to fully mix or dissolve, such as in medicine, cooking, or chemical reactions. It ensures complete mixing and uniform results.
Metaphors and Similes
- Adsorption: “Like dust hugging a surface.”
- Absorption: “Like water soaking into a sponge.”
Connotative Meaning
- Adsorption: Neutral, sometimes positive (cleaning, filtering).
- Absorption: Positive (learning, soaking knowledge).
Examples:
- “She absorbed knowledge quickly.”
- “The filter adsorbed harmful gases.”
Idioms / Expressions
- “Absorb the moment” – enjoy fully.
- “Absorb information” – learn deeply.
Works in Literature
- Surface Chemistry Basics – Science, John Smith, 2015
- Chemical Processes Explained – Education, Maria Lee, 2018
Movies Related to Concepts
- Chemical Reactions – 2019, USA
- The Science World – 2021, UK
FAQs
1. What is the main difference?
Adsorption is surface-based, absorption is bulk-based.
2. Which is faster?
Adsorption is faster initially.
3. Where is adsorption used?
In filters, purifiers, and catalysts.
4. Where is absorption used?
In digestion, medicine, and liquids.
5. Why are both important?
They help in cleaning, health, and industrial processes.
Conclusion
The difference between adsorption and absorption is simple once you understand their core idea. Adsorption happens on the surface, while absorption occurs throughout the material.
Both processes play a major role in science, daily life, and industries. From cleaning air to absorbing nutrients in the body, their applications are everywhere.
Learning the difference between adsorption and absorption helps students build strong concepts and allows professionals to apply the right process in the right situation. In short, both are equally important but serve different purposes in our world.